RESEARCH FOCUS ON DB LAB
The research in the laboratory focuses on developing sustainable and efficient strategies for the construction of carbon–carbon, carbon–oxygen, and carbon–nitrogen bonds, which are central to the synthesis of natural products, complex organic molecules, and pharmaceutically relevant compounds. Emphasis is placed on transition metal catalysis, alongside emerging electro-organic synthesis approaches, to design environmentally benign and versatile methodologies. In particular, we explore the use of earth-abundant and non-precious metals such as nickel (Ni), iron (Fe), and manganese (Mn), often in the form of well-defined pincer complexes, as viable alternatives to traditional precious metal catalysts. These efforts aim to enable highly selective, cost-effective, and sustainable organic transformations with broad applicability in modern synthetic chemistry.
1. Electrocatalysis and its Applications in Organic Synthesis
Electroorganic synthesis is an emerging and sustainable approach in organic chemistry that uses electric current as a clean reagent to drive chemical transformations. By replacing traditional stoichiometric oxidants and reductants, it enables precise control over redox processes, often under mild conditions, while minimizing waste generation. This strategy not only improves functional group tolerance and selectivity but also aligns with green chemistry principles, making it a powerful tool for the efficient synthesis of value-added organic molecules.
- 1. Green Chem. 2024, 26, 5770-5789.
- 2. Chem. Eur. J. 2025, 31, e202501287.
- 3. Green Chem. 2026, 28, 3512-3526.
- 4. Trends in Chem. 2026, Just Accepted.
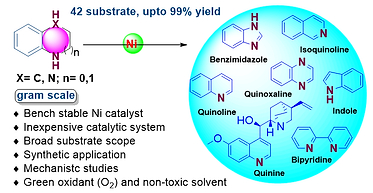
2. Non-precious metal mediated borrowing hydrogen catalysis in the synthesis of C-X (X = C, N, O) bonds
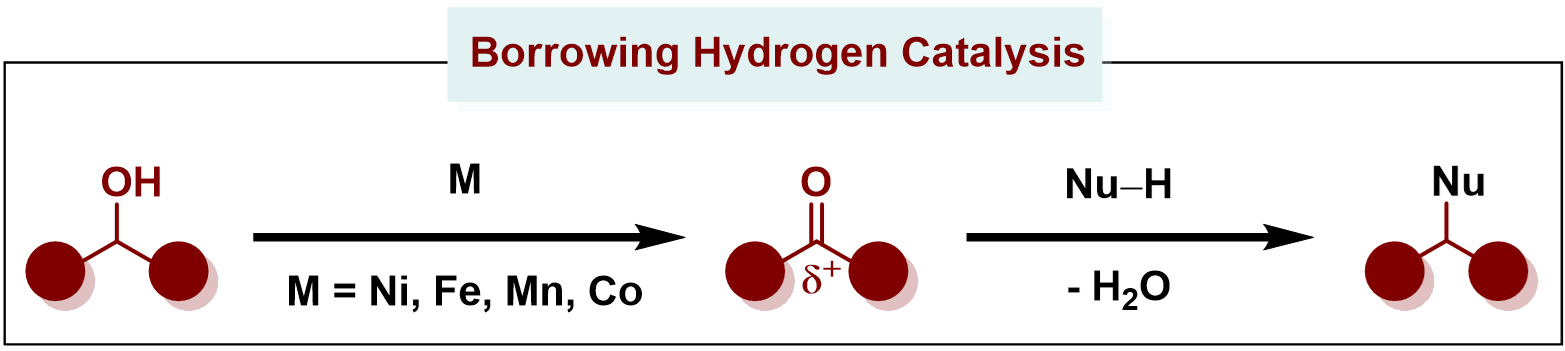
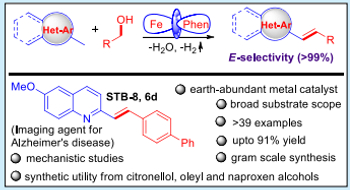
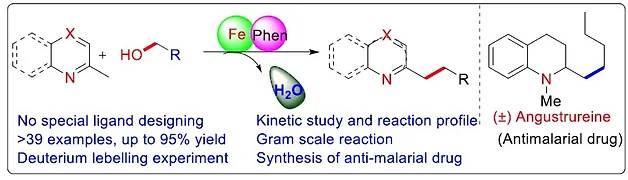
Alcohols are abundant and versatile building blocks in organic chemistry, but their low reactivity often makes direct functionalization challenging without prior activation. Hydrogen Borrowing Catalysis offers an elegant, sustainable solution by enabling redox-neutral transformations of unprotected alcohols into complex molecules, producing only water as a green by-product.
- 1. ACS Catal. 2017, 7, 8152–8158.
- 2. ACS Catal. 2024, 14, 4018-4029.
- 3. Green Chem. 2018, 20, 2250-2256.
- 4. Chem. Soc. Rev. 2024, 53, 4607–4647.
- 5. Chem. Commun. 2025, 61, 19249-19252.
- 6. Org. Lett. 2026, 28,277-282.
- 7. Org. Lett. 2026, 28,522-527.
- 8. Org. Lett. 2024, 26,9299-9304.
- 9. Org. Lett. 2020, 22,6458-6463.
- 10. Org. Lett. 2019, 21,7514-7518.
3. Activation of Small Molecules & Functionalization of Unactivated C-H bonds
- 1. Chem. Commun, 2018, 54, 12369-12372
- 2. Org. Lett, 2019, 21, 157514-187518
- 3. Chem. Commun, 2020, 56, 4777-4780
- 4. Org. Lett, 2020, 22, 6458-6463
5. Heterogeneous catalysis for Sustainable Organic Transformations
- (i) Carbon dioxide to valuable specialty chemicals and bio-fuel
- (ii) Renewable bio-mass to industrially important products
- (iii) De-polymerization of bio-polymers
6. Photoredox-Catalysis and Bio-catalysis
The sustainable route for the construction of new C-X bonds using multiple activation modes to create a complex molecular framework. importantly, used of photo-redox and bio-catalysis avoids the use of multi-step synthesis and streamline the synthetic route
Manuscript submitted (ICMR sponsored project)
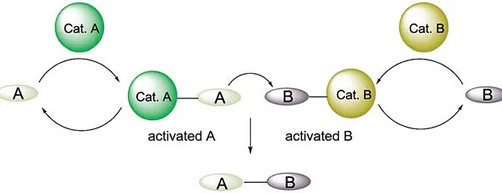
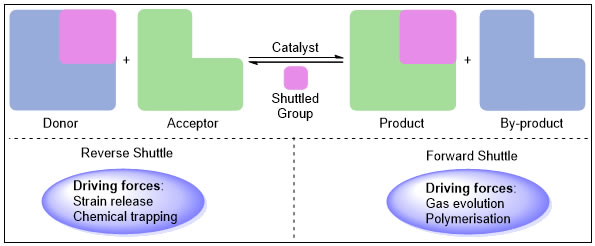
7. Transition metal catalyzed shuttle catalysis:
With the aid of shuttle catalysis, we want to introduce carbonyl, cyano, halides etc. without even using poisonous gases like CO, HCN etc. Thus, it provides a gas free approach to synthesize a number of useful organic entities using surrogates that finds application in the synthesis of naturally product derivatives and pharmaceutically relevant compounds.